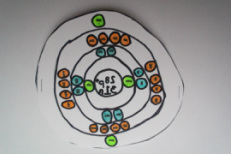
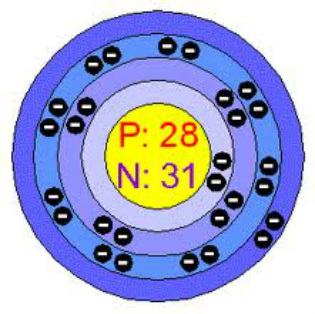
Bohr Model of Nickel Atom
A stable nickel atom has 28 protons, 28 electrons, and 31 neutrons. Protons have a positive charge, neutrons have a neutral charge, and electrons have a negative charge. The nickel atom has 28 protons and 28 electrons so the element stays stable and does not have an overly positive or negative charge.The protons and neutrons are all inside the nucleus and have their atomic mass unit is 1. The electrons float around the nucleus in space and in this case there are 28 of them. Electrons are so small that they have no weight in an atom. My hand-drawn model is on the top and another Bohr model of a nickel atom is on the bottom.